Project
N2O isotope fractionation method

N2O isotope fractionation method to estimate N2 fluxes from soils – methodical development and validation
Denitrification often causes high losses of reactive nitrogen from agricultural soils, but is hardly predictable and up to now difficult to measure in the field. Is it possible to use stable isotopic signatures of N2O from soil to estimate N2 fluxes from denitrification? We tackle this question using controlled process studies in the laboratory and in the field
Background and Objective
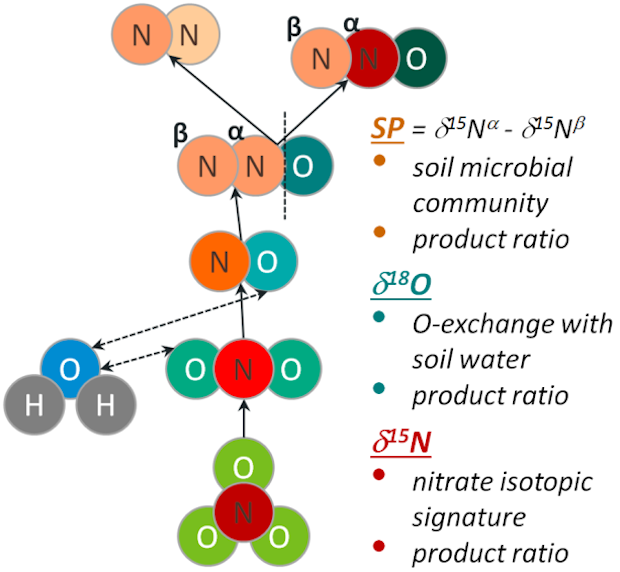
Denitrification reduces the plant-available nitrogen in soils and it is an essential process for the emission of the greenhouse gas N2O. Denitrification represents thus a key process in the N balance, especially in fertilized agricultural ecosystems. Due to methodical limitations in the measurement of N2, denitrification is very difficult to determine at the field scale. The production and reduction of N2O by denitrification and associated isotope effects result in a specific isotopic signature of the N2O molecule, comprising of the relative enrichment or depletion in 15N and in heavy O isotopes (17O, 18O) and of the site specific distribution of 15N within the linear N2O molecule. The isotopic siganture of N2O is controlled by the following variables: the 15N and 17O/18O signatures of precursor compounds (nitrate, soil water), the O-exchange with the soil water during N2O production, the rates of N2O production and N2O reduction to N2 as well as the isotope effects (fractionation) of the various sub-processes. Theoretically, the reduction of N2O to N2 - and consequently the N2 emission - can be derived from the isotopic signature of the emitted N2O, if the other parameters, affecting this signature, can be determined reliably enough. It is our aim to evaluate this methodological approach, i.e. the N2O isotope fractionation method for the determination of N2 emissions, in laboratory experiments with soils.
Approach
The N2O isotope fractionation method for the determination of N2 emissions is tested by laboratory soil incubations. Initially, incubation experiments are carried out to determine one or more control parameters directly. Some of these parameters are determined for the first time (isotope effect of the O-exchange with H2O) or for the first time with a new method (O exchange with H2O based on the 17O anomaly in the produced N2O). An existing isotope fractionation model is expanded in order to simulate and validate the isotopic signature of the emitted N2O based on the parameters determined before. Finally, soil incubation experiments with different soils and varied process conditions are performed with approaches that allow the simultaneous determination of N2O isotopic signatures and the magnitude of the N2 emission. By comparing the measured N2 emissions with the respective values calculated with the isotope fractionation model, it is examined to which extent the N2O fractionation method is suitable for the determination of N2 emissions and provides a new option for the detection of N2 emissions at the field scale.
Our Research Questions
- Is the isotopic fractionation during denitrification stable or predictable? Which factors control the magnitude of isotope fractionation factors?
- Which information can be derived from isotopic signatures of oxygen in N2O and how are they influenced by O-exchange with soil water?
- Can we quantify N2O reduction to N2 based on N2O isotopic signatures? Which requirements have to be taken into account and what is the achievable precision?
- Is the N2O isotopic fractionation method applicable for field studies?
Thünen-Contact

Involved Thünen-Partners
Involved external Thünen-Partners
- Georg-August-Universität Göttingen
(Göttingen, Deutschland)
Funding Body
-
Deutsche Forschungsgemeinschaft (DFG)
(national, öffentlich)
Duration
10.2011 - 3.2015
More Information
Project funding number: DFG We 1904/4-1
Project status:
finished
Publications
- 0
Jung M-Y, Gwak J-H, Rohe L, Giesemann A, Kim J-G, Well R, Madsen EL, Herbold CW, Wagner M, Rhee S-K (2019) Indications for enzymatic denitrification to N2O at low pH in an ammonia-oxidizing archaeon. ISME J 13:2633-2638, DOI:10.1038/s41396-019-0460-6
- 1
Lenhart K, Behrendt T, Greiner S, Steinkamp J, Well R, Giesemann A, Keppler F (2019) Nitrous oxide effluxes from plants as a potentially important source to the atmosphere. New Phytol 221(3):1398-1408, DOI:10.1111/nph.15455
- 2
Ostrom NE, Gandhi H, Coplen TB, Toyoda S, Böhlke JK, Brand WA, Casciotti KL, Dyckmans J, Giesemann A, Mohn J, Well R, Yu L, Yoshida N (2018) Preliminary assessment of stable nitrogen and oxygen isotopic composition of USGS51 and USGS52 nitrous oxide reference gases and perspectives on calibration needs. Rapid Comm Mass Spectrometry 32(15):1207-1214, DOI:10.1002/rcm.8157
- 3
Wu D, Senbayram M, Well R, Brüggemann N, Pfeiffer B, Loick N, Stempfhuber B, Dittert K, Bol R (2017) Nitrification inhibitors mitigate N2O emissions more effectively under straw-induced conditions favoring denitrification. Soil Biol Biochem 104:197-207, DOI:10.1016/j.soilbio.2016.10.022
- 4
Lewicka-Szczebak D, Augustin J, Giesemann A, Well R (2017) Quantifying N2O reduction to N2 based on N2O isotopocules - validation with independent methods (helium incubation and 15N gas flux method). Biogeosciences 14(3):711-732, DOI:10.5194/bg-14-711-2017
- 5
Schorpp Q, Riggers C, Lewicka-Szczebak D, Giesemann A, Well R, Schrader S (2016) Influence of Lumbricus terrestris and Folsomia candida on N2O formation pathways in two different soils - with particular focus on N2 emissions. Rapid Comm Mass Spectrometry 30(21):2301-2314, DOI:10.1002/rcm.7716
- 6
Lewicka-Szczebak D, Dyckmans J, Kaiser J, Marca A, Augustin J, Well R (2016) Oxygen isotope fractionation during N2O production by soil denitrificication. Biogeosciences 13(4):1129-1144, DOI:10.5194/bg-13-1129-2016
- 7
Dyckmans J, Lewicka-Szczebak D, Szwec Lars, Langel R, Well R (2015) Comparison of methods to determine triple oxygen isotope composition of N2O. Rapid Comm Mass Spectrometry 29(21):1991-1996, DOI:10.1002/rcm.7311
- 8
Lewicka-Szczebak D, Well R, Bol R, Gregory AS, Matthews GP, Misselbrook TH, Whalley WR, Cárdenas LM (2015) Isotope fractionation factors controlling isotopocule signatures of soil-emitted N2O produced by denitrification processes of various rates. Rapid Comm Mass Spectrometry 29:269-282, DOI:10.1002/rcm.7102
- 9
Mander Ü, Maddison M, Soosaar K, Koger H, Teemusk A, Truu J, Well R, Sebilo M (2015) The impact of a pulsing water table on wastewater purification and greenhouse gas emission in a horizontal subsurface flow constructed wetland. Ecol Eng 80(Spec. iss.):69-78, DOI:10.1016/j.ecoleng.2014.09.075
- 10
Huang T, Gao B, Hu X-K, Lu X, Well R, Christie P, Bakken LR, Ju X (2014) Ammonia-oxidation as an engine to generate nitrous oxide in an intensively managed calcarous Fluvo-aquic soil. Sci Rep 4:3950, DOI:10.1038/srep03950
- 11
Lewicka-Szczebak D, Well R, Köster JR, Fuß R, Senbayram M, Dittert K, Flessa H (2014) Experimental determinations of isotopic fractionation factors associated with N2O production and reduction during denitrification in soils. Geochim Cosmochim Acta 134:55-73, DOI:10.1016/j.gca.2014.03.010
- 12
Mohn J, Wolf B, Toyoda S, Lin C-T, Liang M-C, Brüggemann N, Wissel H, Steiker AE, Dyckmans J, Szwec Lars, Ostrom NE, Casciotti KL, Forbes M, Giesemann A, Well R, Doucett RR, Yarnes CT, Ridley AR, Kaiser J, Yoshida N (2014) Interlaboratory assessment of nitrous oxide isotopomer analysis by isotope ratio mass spectrometry and laser spectroscopy: current status and perspectives. Rapid Comm Mass Spectrometry 28:1995-2007, DOI:10.1002/rcm.6982
- 13
Mander Ü, Well R, Weymann D, Soosaar K, Maddison M, Kanal A, Lohmus K, Truu J, Augustin J, Tournebize J (2014) Isotopologue ratios of N2O and N2 measurements underpin the importance of denitrification in differently N-loaded riparian alder forests. Environ Sci Technol 48:11910-11918, doi:10.1021/es501727h
- 14
Lewicka-Szczebak D, Well R, Giesemann A, Rohe L, Wolf U (2013) An enhanced technique for automated determination of 15N signatures of N2, (N2+N2O) and N2O in gas samples. Rapid Comm Mass Spectrometry 27(13):1548-1558, DOI:10.1002/rcm.6605
- 15
Giesemann A, Lewicka-Szczebak D, Well R (2013) Automated analysis of 15N signatures of N2 (N2 + N2O) and N2O in gas samples - advances in measurement technique : [Abstract for] Cost-Sibae 2013 Meeting: Challenges in the Applications of Stable Isotopes across Disciplines and Scales, Wroclaw, Poland; 05/2013.
- 16
Lewicka-Szczebak D, Senbayram M, Köster JR, Well R (2013) Isotope fractionation factors of N2O production and reduction by denitrification: b. Modeling data from soil incubation under N2-free atmosphere. Geophys Res Abstr 15:8864
- 17
Well R, Weymann D, Lewicka-Szczebak D, Rohe L, Flessa H (2013) Isotope fractionations factors of N2O production and reduction by denitrification: a. Laboratory incubation studies using N2O reductase inhibition. Geophys Res Abstr 15:8210
- 18
Köster JR, Well R, Tuzson B, Bol R, Dittert K, Giesemann A, Emmenegger L, Manninen A, Cárdenas LM, Mohn J (2013) Novel laser spectroscopic technique for continuous analysis of N2O isotopomers - application and intercomparison with isotope ratio mass spectrometry. Rapid Comm Mass Spectrometry 27(1):216-222, DOI:10.1002/rcm.6434
- 19
Mukotaka A, Toyoda S, Yoshida N, Well R (2013) On-line triple oxygen isotope analysis of nitrous oxide using decomposition by microwave discharge. Rapid Comm Mass Spectrometry 27(21):2391-2398, DOI:10.1002/rcm.6698
- 20
Köster JR, Well R, Dittert K, Giesemann A, Lewicka-Szczebak D, Mühling KH, Herrmann A, Lammel J, Senbayram M (2013) Soil denitrification potential and its influence on N2O reduction and N2O isotopomer ratios. Rapid Comm Mass Spectrometry 27(21):2363-2373, DOI:10.1002/rcm.6699
- 21
Köster JR, Senbayram M, Dittert K, Well R, Giesemann A, Lewicka-Szczebak D, Mühling KH (2013) Soil denitrification potential and its influence on the N2O / N2 product ratio and N2O isotopomer ratios. Geophys Res Abstr 15:13171
- 22
Lewicka-Szczebak D, Senbayram M, Köster JR, Well R (2013) Stable isotopic signatures of N2O produced by denitrification: Experimental determination of fractionation factors : [Abstract for] Cost-Sibae 2013 Meeting: Challenges in the Applications of Stable Isotopes across Disciplines and Scales, Wroclaw, Poland; 05/2013.
- 23
Well R, Eschenbach W, Flessa H, Heide C von der, Weymann D (2012) Are dual isotope and isotopomer ratios of N2O useful indicators for N2O turnover during denitrification in nitrate-contaminated aquifers? Geochim Cosmochim Acta 90(1):265-282, DOI:10.1016/j.gca.2012.04.045

![[Translate to English:] [Translate to English:]](/media/_processed_/6/4/csm_titel_CO2Kampagne8_afeea2273e.png)
![[Translate to English:] [Translate to English:]](/media/_processed_/4/1/csm_titel_93px_CO2Kampagne8_9b0f3354d4.png)





